Publication Date: Wed Nov 08 00:00: Research Org.: Energy Frontier Research Centers (EFRC) (United States).

Reactions between O and other species tend to be more prevalent than reactions more » between O 2 and other species, particularly on more reactive surfaces. More open surfaces or undercoordinated defect sites can be much more reactive than close-packed surfaces.
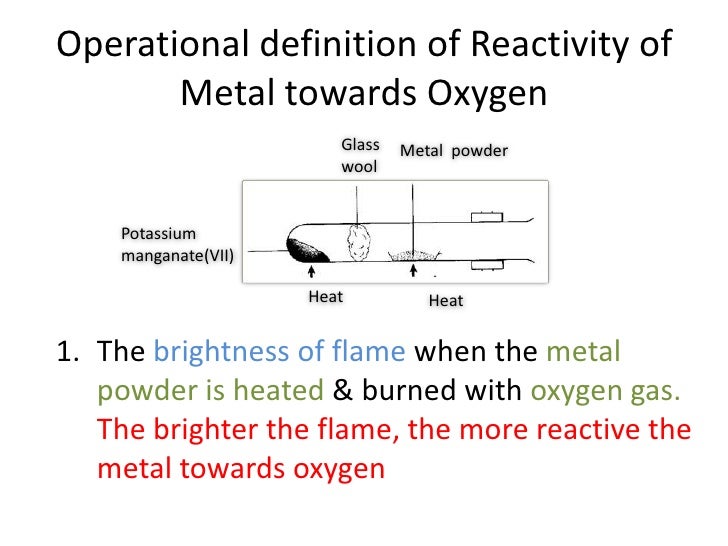
#Reactivity of oxygen full
Periodic trends can be rationalized in terms of attractive and repulsive interactions with the d-band, such that inert metals tend to feature a full d band that is low energy and has a large spatial overlap with adsorbate states. The reactivity of a surface toward O 2 generally correlates with the adsorption strength of O, the tendency to oxidize, and the heat of formation of the oxide. The reactivity of O 2 and O toward reactant molecules is also briefly discussed in the context of catalysis. The mechanism and dynamics of O 2 dissociation are also reviewed, including the importance of the spin transition. Here, trends in the activation of O 2 on transition metal surfaces are discussed, and various O 2 adsorption states are described in terms of both electronic structure and geometry. Fundamental studies of well-defined metal surfaces using a variety of techniques have given crucial insight into the mechanisms, energetics, and dynamics of O 2 adsorption and dissociation.
The activation of O 2 on metal surfaces is a critical process for heterogeneous catalysis and materials oxidation.
